Una solución para la adquisición de medicamentos: Cómo Brasil está mejorando la toma de decisiones mediante datos abiertos e inteligencia artificial

Desafío
Más de 150 millones de personas dependen del sistema de salud universal de Brasil, que promete acceso gratuito a medicamentos esenciales. Sin embargo, la adquisición de medicamentos está fragmentada con miles de gobiernos locales adquiriendo productos en sistemas dispersos, lo que genera grandes variaciones de precios y documentación inconsistente. Sin información confiable, los compradores públicos no pueden tomar decisiones inteligentes y los ciudadanos pagan más. La supervisión es casi imposible, incluso para los 15 millones de brasileños que reportan no recibir los medicamentos que necesitan.
Enfoque de contratación abierta
Una coalición de múltiples partes interesadas, que incluye a Transparência Brasil, la Contraloría General de la República y el Ministerio de Gestión e Innovación, trabajó para lograr que los precios de los medicamentos sean más transparentes y comparables, y para reducir el costo de suministro en un mercado que, por lo demás, es volátil y opaco. Comenzaron construyendo una nueva plataforma, llamada Medicamentos Transparentes, para que los compradores obtengan precios de referencia claros para medicamentos vitales, que fue desarrollada con el apoyo del programa Lift de la OCP. La plataforma aprovecha un amplio modelo de lenguaje para depurar, estandarizar y clasificar los datos del Portal Nacional de Contrataciones Públicas (PNCP), estructurados según el Estándar de Datos para las Contrataciones Abiertas (OCDS). La plataforma agrega, por primera vez, las compras de medicamentos realizadas por los más de 5.000 municipios, 27 estados y entidades federativas del país a través de 263 sistemas diferentes. La herramienta se probó y mejoró en colaboración con el estado de Minas Gerais y el municipio de São Paulo, así como con actores clave como el Tribunal de Cuentas de la Unión.
Resultados
Los funcionarios públicos han comenzado a utilizar la nueva plataforma, Medicamentos Transparentes, para realizar comparaciones de precios que les ayudan a tomar decisiones más acertadas y justas. También, sirve como referencia para la compra de medicamentos, según lo exige la ley de contrataciones públicas. Las capacitaciones para municipios y organismos de control promueven y garantizan su uso.
La plataforma y la interacción en torno a ella brindarán nuevas oportunidades para el seguimiento de licitaciones exitosas, la recopilación de datos clave sobre la evolución de los precios a lo largo del tiempo y la mejora de la definición de precios de referencia. Continuaremos trabajando con el gobierno y las organizaciones de la sociedad civil para integrar la plataforma en un proceso de toma de decisiones práctico y basado en datos, de modo que sirva a los millones de brasileños que necesitan acceso confiable a medicamentos.
Cada año se gastan 75 mil millones de dólares en medicamentos a través del sistema de salud universal de Brasil. Sin embargo, 15 millones de brasileños aún reportan no recibir el tratamiento que necesitan. El acceso varía significativamente en diferentes partes del país. Al norte de Brasil, una zona más rural, los residentes pagan más por medicamentos esenciales a pesar de tener ingresos más bajos.
Una iniciativa conjunta entre el gobierno y la sociedad civil, apoyada por el programa Lift de OCP, está ayudando a mejorar la gestión basada en datos de las compras de medicamentos en Brasil. A largo plazo, esta reforma podría sentar las bases para fortalecer la supervisión de un sector complejo y de alto valor, y empoderar a los pacientes para que reciban la atención a la que tienen derecho.
Gracias a esta colaboración, la adquisición de productos médicos en todo el país pueden compararse en un único sitio web gratuito. Con más de 1500 visitantes después del primer mes, la plataforma ahora está siendo utilizada por los funcionarios de compras públicas para crear comparaciones de precios que les ayudan a tomar decisiones más acertadas y justas.
En un momento en que Brasil está redefiniendo su estrategia nacional de compras, el proyecto proporciona un modelo sobre cómo el país puede aprovechar las compras públicas para diseñar e implementar mejores políticas públicas.
Desarrollo de Medicamentos Transparentes: Desbloqueo de datos vitales para tomar mejores decisiones
Desde su lanzamiento en abril de 2025, Medicamentos Transparentes publica las compras de medicamentos realizadas por todas las entidades gubernamentales a nivel nacional y subnacional durante los últimos dos años, lo que permite comparar el precio de los medicamentos entre regiones y a lo largo del tiempo, basándose en características como ingredientes activos, dosis, método de adquisición, cantidad y proveedor. El diseño simple del portal es la culminación de un importante trabajo interno para reunir toda esta información en un solo lugar.
Dado que las compras públicas en Brasil están descentralizadas, las transacciones y procesos de cada estado y municipio se documentan en miles de portales de transparencia y formatos de datos diferentes, lo que dificulta el monitoreo sistemático. Esta falta de datos interoperables crea un terreno fértil para la ineficiencia y la mala gestión, problemas de presupuesto y suministro, e impide que la sociedad civil y los periodistas rastreen las compras o detecten sobreprecios o corrupción.
Fortalecer la transparencia y el seguimiento de las compras de medicamentos es fundamental para garantizar el derecho al acceso a la salud.
“Fortalecer la transparencia y el monitoreo de las compras de medicamentos es fundamental para garantizar el derecho al acceso a la salud”, afirmó Juliana Sakai, Directora Ejecutiva de Transparência Brasil.
En 2021, Brasil aprobó una nueva ley de contratación pública para modernizar la contratación pública y mejorar la transparencia y eficiencia del sistema. La legislación exige que los 5.568 municipios, 27 estados y entidades federativas divulguen su información de contratación, a lo largo de todo el ciclo de contratación, en una plataforma centralizada: el Portal Nacional de Contrataciones Públicas . La ley también encomienda al gobierno la creación de una base de datos de precios de productos sanitarios para transparentar los patrones de costos de los tratamientos y promover una contratación más eficiente en un sector que supera con creces a la mayoría de los demás mercados en cuanto a número de compras y gasto total. Sin embargo, la falta de estandarización de datos en los 263 sistemas existentes para la compra y documentación de las compras farmacéuticas en los estados y municipios ha sido un obstáculo para la implementación de dicho «banco de precios».
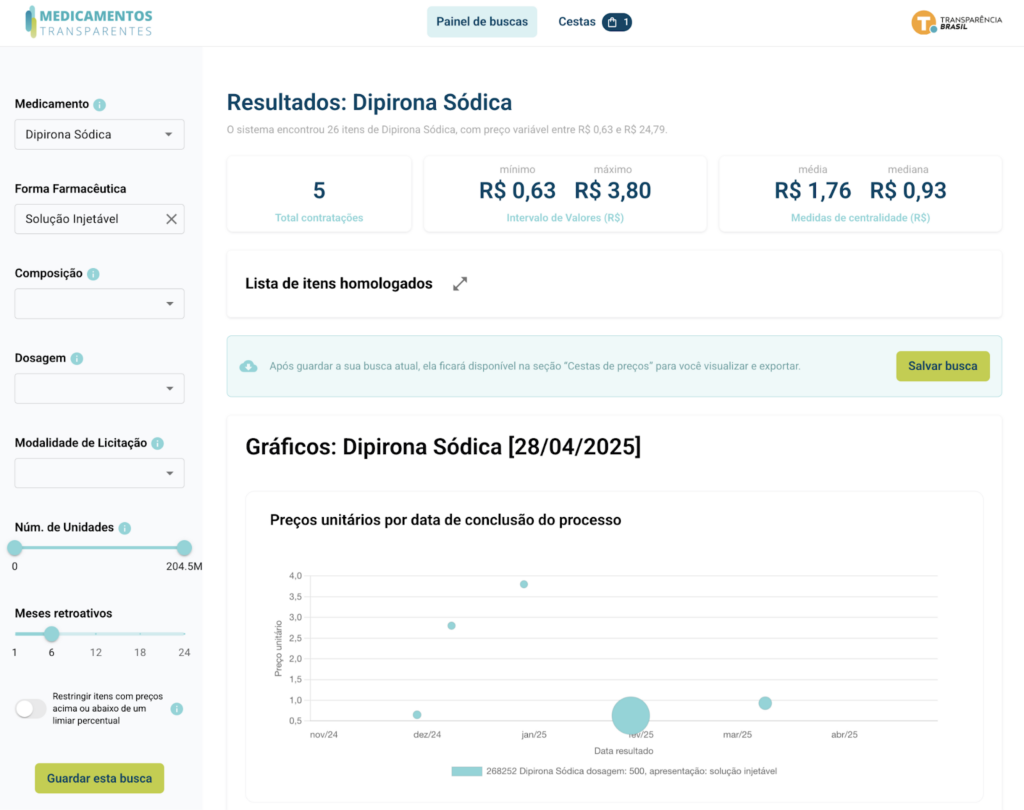
Medicamentos Transparentes resuelve este problema utilizando datos abiertos del PNCP y del Sistema de Catálogo de Materiais e Serviços do Governo Federal (CATMAT/CATSER), empleando la inteligencia artificial para clasificar los artículos como medicamentos y estructurándolos en el Estándar de Datos para las Contrataciones Abiertas (OCDS) para que puedan compararse con precisión. Fue diseñado para ayudar a los gestores y funcionarios públicos a garantizar una mayor eficiencia en las compras públicas de medicamentos.
Lucas Pedersoli, quien lidera la investigación de precios de compras en el estado de Minas Gerais, destaca el valor del portal para la planificación de compras, así como para establecer un vocabulario común para las compras farmacéuticas en todo el país.
“La plataforma ha sido increíblemente útil. Los filtros que tiene reducen el tiempo de búsqueda y todos los medicamentos se encuentran en un único catálogo de medicamentos en lugar de tener varios. Los datos proporcionados no solo nos ayudaron con la investigación de precios, sino también con otros componentes de la investigación de mercado que son igualmente importantes para el éxito del proceso”.
Medicamentos Transparentes fue desarrollado con el apoyo del programa acelerador de impacto Lift de la OCP, por una coalición de socios que incluye autoridades de compras y fiscalización, así como la sociedad civil. Transparência Brasil, organización independiente con 25 años de experiencia en la promoción de un mejor gasto público, colaboró con personal de SEGES, responsable del sistema federal de compras electrónicas y administrador del portal de transparencia del PNCP.
Su trabajo se llevó a cabo con el estrecho apoyo del Director de Investigación de Precios de la Subsecretaría de Compras Públicas del Estado de Minas Gerais y de los principales organismos reguladores, la Contraloría General de la República (CGU) y el Tribunal de Cuentas de la Unión (TCU), quienes compartieron las lecciones de un proyecto anterior centrado en el análisis de la adquisición de medicamentos a partir de datos de facturas (Análisis de Precios de Artículos de Licitación, o APRIL). Para comprender mejor los puntos críticos, también se contó con la participación de compradores públicos subnacionales, como el estado de Minas Gerais y el municipio de São Paulo.
El objetivo inicial del equipo era reducir el costo de los medicamentos con sobreprecio hasta en un 20%. Sin embargo, poco después de comenzar el trabajo, se dieron cuenta de lo difícil que era obtener precios unitarios y crear una canasta de precios consistente que les permitiera establecer una línea de base y monitorear estos procesos. Por lo tanto, reorientaron su enfoque para lograrlo primero.
Un enfoque colaborativo de contratación abierta
Mejorar la accesibilidad y la calidad de los datos de adquisición de medicamentos fue un paso esencial para que la adquisición de estos fuera más justa y eficiente. Permitiría al equipo realizar un análisis comparativo de las compras de medicamentos, identificando los costos típicos de los tratamientos y las anomalías. Datos más precisos podrían aumentar la eficiencia de las entidades contratantes a la hora de determinar precios de referencia y optimizar la relación calidad-precio. También, permitiría una supervisión más eficaz y un seguimiento automatizado, con un alto grado de precisión para identificar irregularidades e ineficiencias en la compra de medicamentos.
A pesar de que el sistema fuente pública datos abiertos a través de una API, las limitaciones de disponibilidad y usabilidad dificultan la búsqueda de los detalles necesarios para realizar el análisis de precios. Por ejemplo, en lugar de estar estructurados en campos de datos separados, los detalles relevantes a menudo se encuentran en campos de texto libre. En un caso, un comprador público solo incluyó el nombre del principio activo del medicamento, sin detallar su concentración, origen, dosis ni otras características relevantes. Si bien la información completa puede estar disponible en el PDF de la licitación o en otros documentos, estos formatos no son legibles por máquina. A lo largo del proyecto, se realizaron varias sesiones de trabajo en las que el equipo de Transparência Brasil presentó sus hallazgos sobre la usabilidad, calidad y cobertura de los datos abiertos del PNCP, junto con propuestas de solución.
Por ejemplo, Transparência Brasil realizó un análisis de la calidad de los datos de las compras de medicamentos e identificó deficiencias clave en las descripciones de las compras registradas en la base de datos del PNCP que dificultaban la comparación de precios. Se encontró que el 30% de los procesos de adquisición de medicamentos presentaban descripciones insuficientes o incorrectas. Los informes se presentaron a SEGES, que implementó varias recomendaciones (primer informe, segundo informe, tercer informe), incluyendo el 44% (11 de 25 recomendaciones) relacionadas con la usabilidad, el 30% (3/10) con el acceso a los datos y la infraestructura, y el 66% (3/5) con la calidad de los datos.
“El análisis y la retroalimentación proporcionados a través de estos informes fueron sumamente valiosos para la SEGES, ya que nos permitieron identificar rápidamente oportunidades concretas de mejora en el PNCP. Contar con información basada en evidencia nos ayudó a responder con mayor eficiencia, priorizar las acciones correctivas y centrar nuestros esfuerzos en las funcionalidades y los elementos de datos que los usuarios realmente necesitan para un monitoreo y análisis efectivos”, afirma Everton Batista dos Santos, Director de Normas y Sistemas de la Secretaría de Gestión e Innovación (SEGES), perteneciente al Ministerio de Gestión e Innovación en los Servicios Públicos (MGI).
Otras partes interesadas participaron en talleres para garantizar que la nueva plataforma satisficiera las necesidades de quienes más probablemente la utilizarían. Entre los asistentes se encontraban compradores públicos subnacionales del Estado de Minas Gerais y del Municipio de São Paulo, quienes demostraron desde el principio su disposición a colaborar, así como monitores de la sociedad civil y periodistas.
“Cuando aumenta el gasto en salud, es fundamental contar con herramientas que faciliten el monitoreo y la eficiencia de este gasto”, destaca Flávia Schmitt, exdirectora de Gobierno Abierto y Transparencia de la CGU. “Y es fundamental que podamos contar con mejoras continuas, colaboración e investigación en torno a estas herramientas para promover políticas públicas más efectivas”.
Uso de IA/LLM para extraer mejor información
Tras la recopilación de datos, es necesario depurarlos y estandarizarlos. La plataforma Medicamentos Transparentes recopila datos mensuales del PNCP, filtra las compras de medicamentos y, a continuación, utiliza un modelo de lenguaje grande (LLM) para asociar cada artículo del PNCP adquirido con el código de producto correspondiente mediante un sistema de clasificación aplicado a las compras federales en Brasil (Catálogo de Materiales o CATMAT Clase 6505), basándose en la similitud entre las descripciones.
Con este modelo, el 80% de los artículos se asociaron correctamente con los artículos CATMAT, en comparación con sólo el 5% durante la revisión inicial de los datos del PNCP realizada por el equipo. Una persona tarda 90 minutos en clasificar 100 artículos, el modelo puede hacerlo en tan solo 5 segundos. Mejorar la calidad de los datos, como recomienda Transparência Brasil, puede aumentar aún más la cantidad de artículos que se pueden clasificar.
Medicamentos Transparentes también brinda acceso abierto a los datos de adquisiciones, incluyendo contratos, artículos y resultados de adjudicaciones, los cuales pueden ser descargados en formato JSON y CSV (también disponibles a través del Registro de Datos de OCP).
Cada descarga contiene datos de adquisiciones mensuales y estatales. Los datos se actualizan mensualmente mediante API públicas y están disponibles bajo una licencia Creative Commons.
Un punto de acceso único a los datos de contrataciones para impulsar el cumplimiento de los objetivos económicos y sociales de Brasil.
El proyecto de transparencia en la contratación de medicamentos orientó esfuerzos más amplios para mejorar la contratación pública mediante la digitalización. En otoño de 2025, SEGES comenzó a publicar datos abiertos estandarizados en el Estándar de Datos para las Contrataciones Abiertas desde sus API en Compras.gov.br, el sistema federal obligatorio de contratación electrónica.
La publicación incluye datos para identificar políticas de sostenibilidad y la participación de las pequeñas empresas en la contratación pública. Por primera vez, toda la información sobre licitaciones y adjudicaciones que se procesan a través del sistema Compras.gov.br está disponible en un formato estructurado, abierto y comparable internacionalmente. Todos los detalles de un proceso de contratación se pueden obtener desde un único punto de acceso, mediante un identificador único.
“Medicamentos Transparentes surgió de la creación del Portal Nacional de Contrataciones Públicas, gracias a su enorme potencial, que reúne datos de todo el país sobre contrataciones públicas. No genera tantos titulares, pero sí un gran impacto”, destaca Juliana Sakai, de Transparência Brasil.
La nueva plataforma de transparencia en medicamentos ha demostrado que contar con datos de contrataciones de alta calidad y listos para el análisis es fundamental para implementar reformas que generen ahorros y mejoren la prestación de servicios públicos esenciales.
Para otros sectores, demuestra que, mediante un enfoque de contratación abierta, es posible lograr comparabilidad en las contrataciones entre regiones y a lo largo del tiempo, incluso en mercados grandes y altamente descentralizados.
El lanzamiento de Medicamentos Transparentes es solo el comienzo. Sus creadores ahora tienen grandes planes para promover el uso de esta herramienta entre gestores públicos, organismos de control, la sociedad civil, periodistas y todos aquellos que desean mejorar la salud pública en Brasil.
En un país donde millones de personas aún tienen dificultades para acceder a tratamientos esenciales, contar con mejores datos puede parecer una solución técnica. Pero cuando los precios son más justos, los mercados son transparentes y las decisiones se basan en información, el impacto puede ser profundamente humano. Al convertir los registros de compras fragmentados en información procesable, los datos abiertos y la IA no son solo herramientas para la eficiencia, sino también instrumentos para la equidad.
Desde OCP estamos entusiasmados de contribuir a este interesante proyecto; capturaremos y compartiremos lecciones a lo largo del recorrido, incluso cómo los nuevos datos informan una mejor compra de medicamentos y si los precios están bajando para los ciudadanos.